By the end of the course, the student should be able to:
-
Knowledge:
Recall key definitions, laws, models, and formulas related to molecular electronic structure, molecular orbitals, reactivity, and conjugated systems. -
Comprehension:
Explain concepts such as Lewis structures, VSEPR geometries, molecular orbitals, frontier orbitals, and aromaticity in their own words. -
Application:
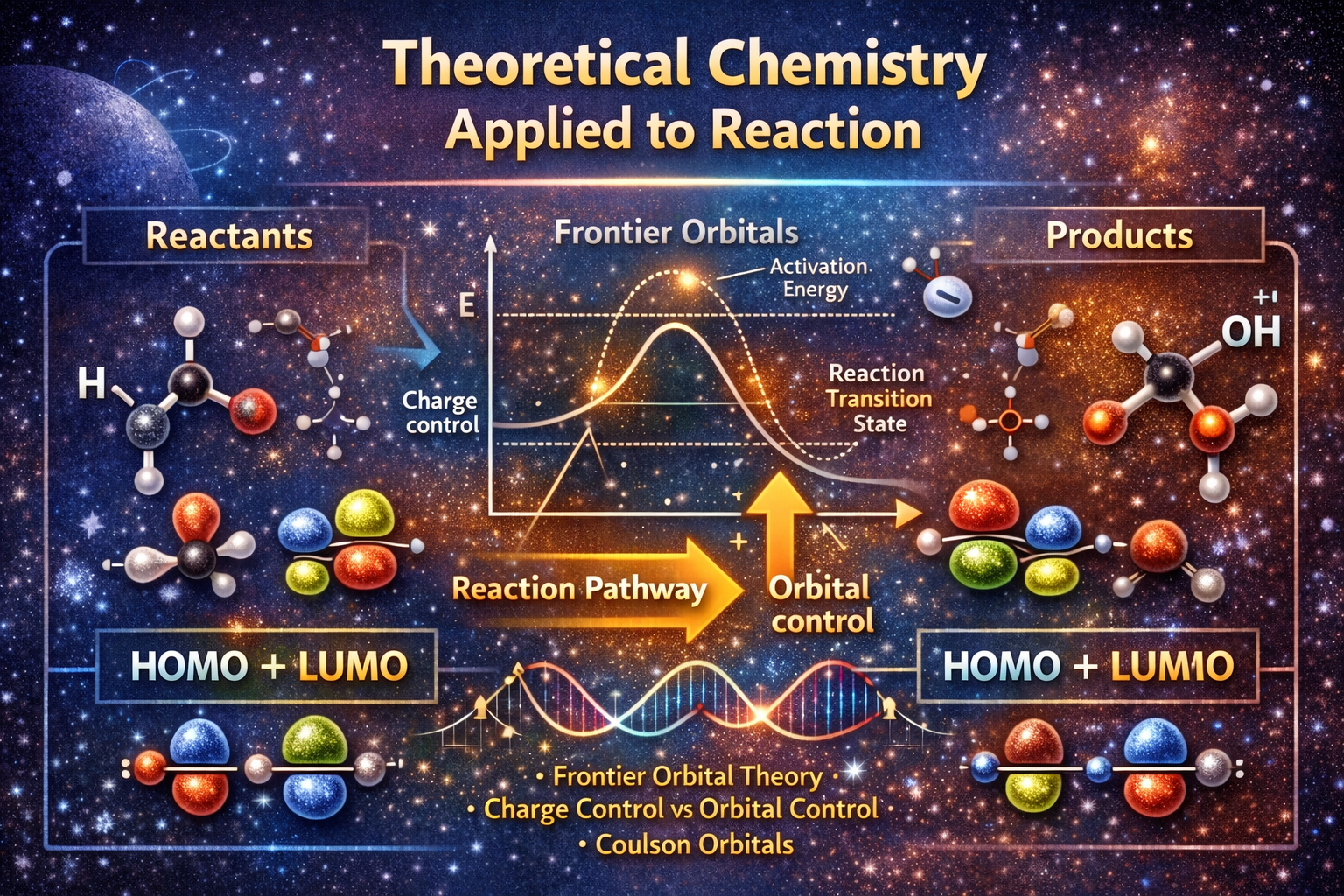
Apply theoretical models (VSEPR, CLOA, Frontier Orbital Theory, Hückel method) to real molecules and simple chemical reactions. -
Analysis:
Analyze molecular structure–reactivity relationships using orbital interactions and electronic effects. -
Synthesis:
Combine different theoretical approaches (Lewis, MO theory, reactivity models) to predict molecular properties and reaction outcomes.
- Enseignant: Sakina HAYAHEME
